SETTING THE GOLD STANDARD FOR CLINICAL DATA AND BIOSPECIMEN
Highest quality biospecimen make all the difference
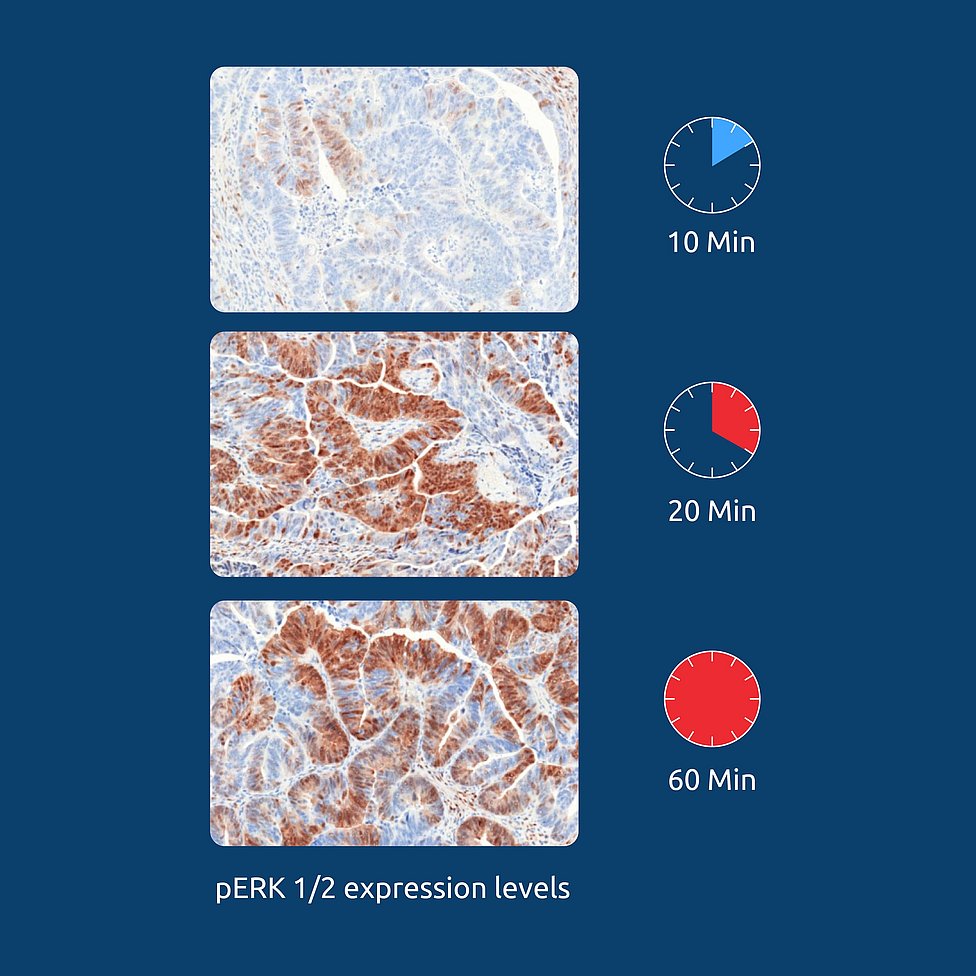
We are convinced that biospecimen quality along with the resulting data is crucial to making all relevant details of a cancer disease transparent. That’s why we apply numerous detailed standard operating procedures (SOPs) to the collection and processing of our wide variety of biospecimen and corresponding clinical data, achieving an ischemia time for tissue of ≤12 minutes and covering around 300 individual data points.
THE INDIVUMED
GLOBAL CLINICAL NETWORK
To ensure a constantly high quality of specimen and the highest level of comparability, all our clinical partners around the globe have implemented our detailed and in-depth SOPs. The procedures have been developed and refined through decades of experience working side-by-side with surgeons and pathologists in our network.
Collaborating with Indivumed Therapeutics
Would you like to become part of our Clinical Network? Are you interested in a collaborative partnership or a career at Indivumed? If so, we look forward to hearing from you.